|
3/22/2023 0 Comments Glims pharmaWhen substituting Glimepride for other such agents, the initial daily dose is 1 mg this applies even in changeover from the maximum dose of other oral blood sugar lowering agents. To avoid hypoglycaemia, timely dose reduction or cessation of Glims therapy must be considered.A dose adjustment must also be considered whenever the patient's weight or life style changes, or other factors arise which cause an increased susceptibility to hypo- or hyperglycaemia.Ĭhangeover from other oral antidiabetics to Glims: There is no exact dosage relationship between Glims and other oral blood sugar lowering agents. Secondary dosage adjustment: As the control of diabetes improves, sensitivity to insulin increases therefore, Glims requirement may fall as treatment proceeds. It is very important not to skip meals after taking the drug. This should be taken immediately before a substantial breakfast or - if none is taken - immediately before the first main meals. Normally, a single daily dose is sufficient. Any increase can be based on regular blood sugar monitoring, and should be gradual, i.e., at intervals of one to two weeks and carried out step wise at follows: 1 mg-2 mg-3 mg-4 mg-6 mg.ĭose range in patients with well controlled diabetes: Usual dose range in patients with well controlled diabetes is 1 to 4 mg daily.ĭistribution of doses: Timing and distribution of doses are decided by the physician, in consideration of the patient's current life style. If necessary, the daily dose can be increased. Initial dose and dose titration: Usual initial dose is 1 mg once daily. Monitoring of glucose levels in blood and urine also serves to detect either primary or secondary failure of therapy. The initial and the maintenance doses are set based on the results of regular checking of glucose in blood and urine. The dosage of Glims must be the lowest which is sufficient to achieve the desired metabolic control. Support any other activities as requested by Manager.In principle, the dosage of Glims is governed by the desired blood sugar level. Drive continuous improvements and lean activities. Support software issues/troubleshooting as needed. Adding/removing instruments, creating/modifying methods, adding/removing sample reports. Administration of Empower and LabX activities. Support daily operation of the Empower, GLIMS, LabX systems. Pay close attention to the work of others and proactively step in to help as needed. Build knowledge of the company, process, and customers. Solve complex problems and take new perspective using existing solutions. Highly visible role requiring attention to detail, self-motivation and organization. The successful candidate will have the ability to work Independently with minimal guidance. The position isn’t LIMS, but Empower to apply knowledge of internal/external business challenges to improve products and processes, and assist in the roll-out of Laboratory Information Systems. Previous experience in a laboratory setting * Hours: 8:00am – 5:00pm (matches Start/End Time in table below?) * Preferred Skills: Preferred Skills: GLIMS, Empower, MIDAS, Trackwise, LabX. * Attention to detail with focus on spelling is a must. * Applies regulatory / compliance requirements, including GMP, NDA, and related regulations, and can interpret those regulations for implementation in workplace. * Capability for continuous product process improvement. * Routine use of analytical tools to solve issues and simplify processes. * MS Office (Excel, Project), GCM, GLIMS and SAP * Proficiency in computer builds and aptitude with information systems. * Minimum 1 year of Empower admin experience. * Required Skills: 2-4yrs industry experience, pharmaceutical industry preferred. 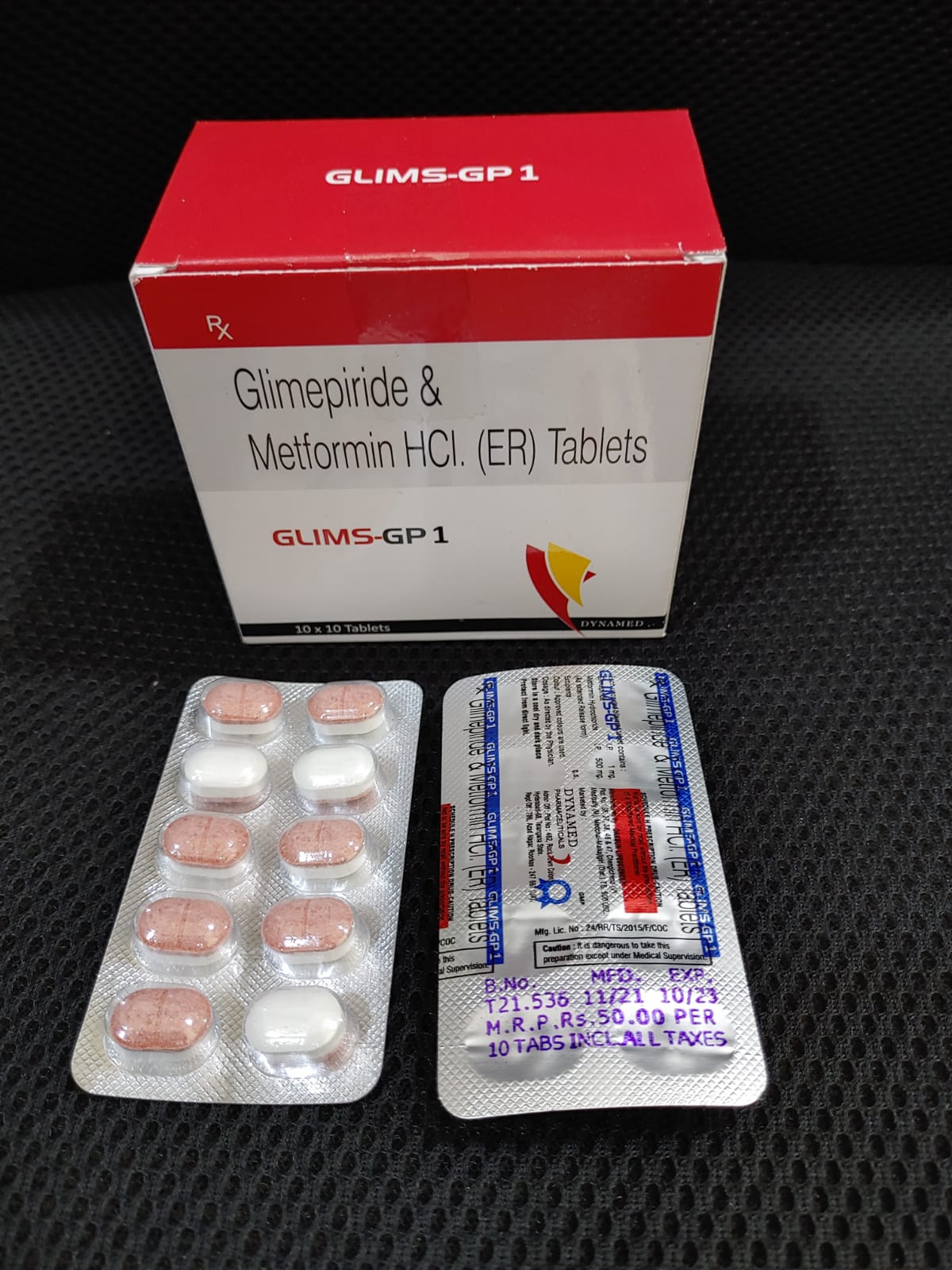
degree (preferably in Science or Engineering), no Ph.D.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
